Answer the question number 14 a by an expert which i easely understand (4) (c) Calculate rate constant of the - Chemistry - Surface Chemistry - 13294039 | Meritnation.com

Two moles of an ideal gas at 2 bar and 27^∘C expand isothermally against a constant pressure of 1 bar. The work done by the gas is equal to :

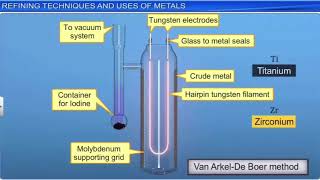
In van Arkel method, if `I_(2)` is introducted at `1800K` anode impure zironium metal, the product - YouTube
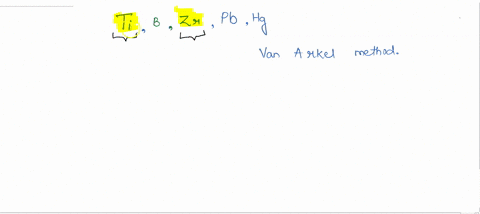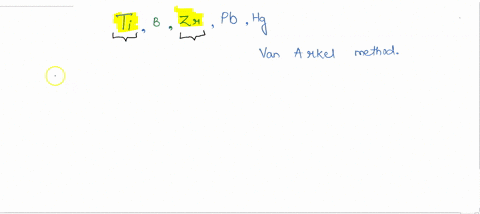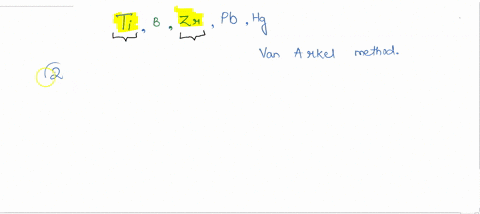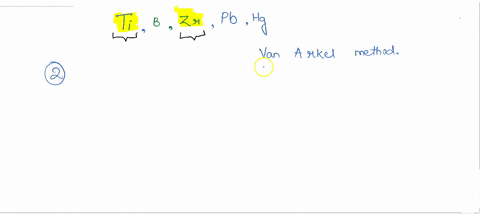
SOLVED:How many metals are commercially purified by Van Arkel method from the given metals? Ti, B, Zr, Pb, Hg

i T i is purified by Van Arkel method. In it, the formula of metal iodide vapour formed is T iI p.ii The oxidation state of F e in Fool's gold is

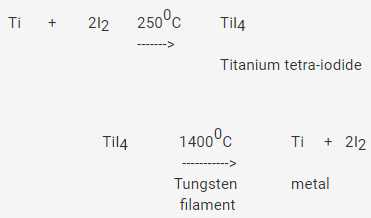
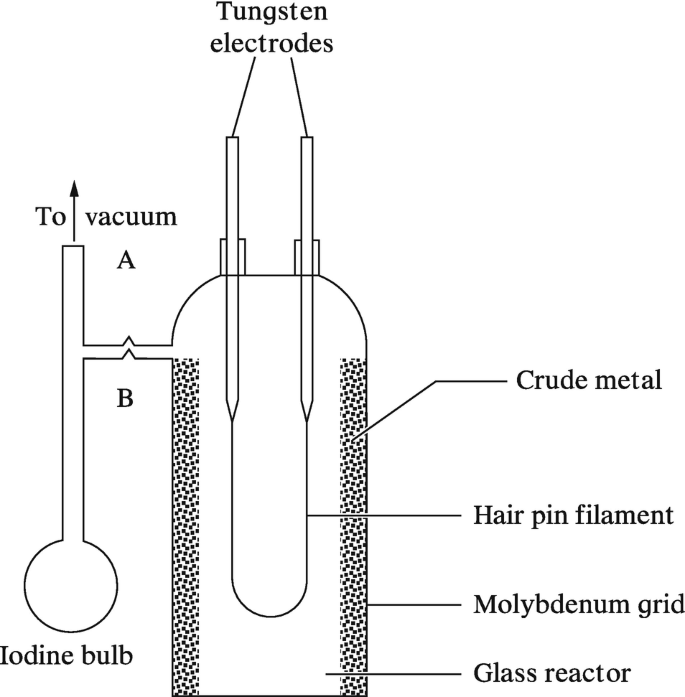
Write Chemical Reactions Involved in Van Arkel Method for Refining Titanium - Chemistry | Shaalaa.com

Respected Ma'am what is zone refining and van arkel method Actually I didn't understand it - Chemistry - - 15658505 | Meritnation.com


![PDF] A Quantitative van Arkel Diagram | Semantic Scholar PDF] A Quantitative van Arkel Diagram | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aa5b5fb921e63b9e37aa523442fd0fd6049d6b74/2-Figure3-1.png)