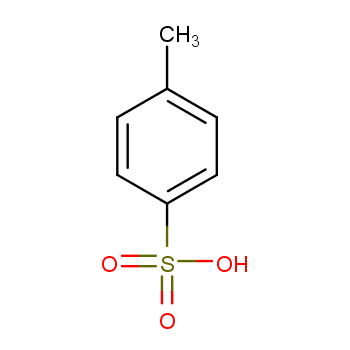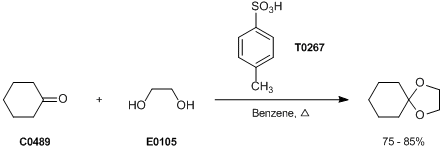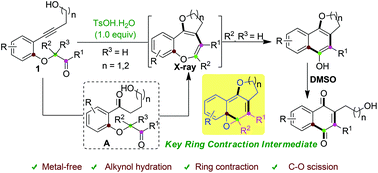
p-TsOH promoted synthesis of benzo-fused O-heterocycles from alkynols via ring contraction and C–O scission strategy - Green Chemistry (RSC Publishing)

Catalytic systems containing p-toluenesulfonic acid monohydrate catalyzed the synthesis of triazoloquinazolinone and benzimidazoquinazolinone derivatives | SpringerLink

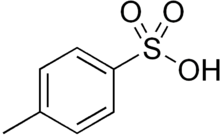
Detailed Characterization of p-Toluenesulfonic Acid Monohydrate as a Convenient, Recoverable, Safe, and Selective Catalyst for Alkylation of the Aromatic Nucleus | The Journal of Organic Chemistry

Catalytic systems containing p-toluenesulfonic acid monohydrate catalyzed the synthesis of triazoloquinazolinone and benzimidazoquinazolinone derivatives | SpringerLink

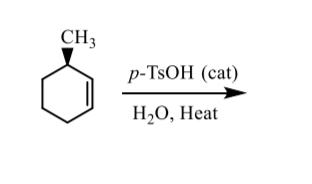
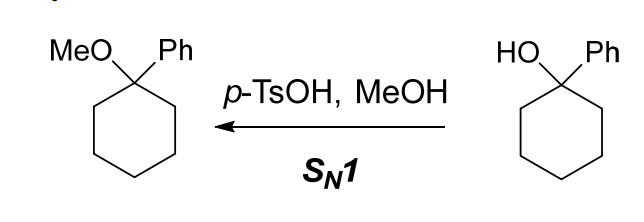
Draw the product formed when the given alcohol undergoes dehydration with TsOH, and label the major product when a mixture results. | Homework.Study.com

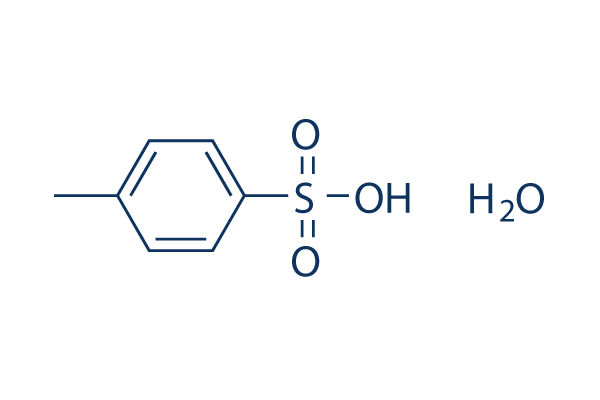
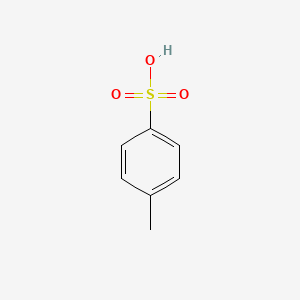
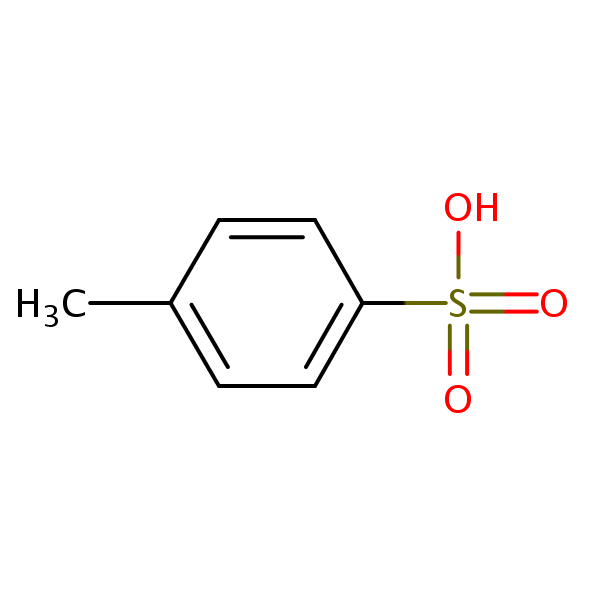
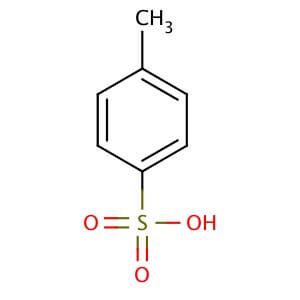
p-Toluenesulfonic Acid Hydrate | 4-Methylbenzenesulfonic Acid Hydrate; 4-Toluenesulfonic Acid Hydrate; Tosic Acid Hydrate; PTSA Hydrate; p-Toluenesulfonic Acid Hydrate; TsOH; p-tsa; Lisinopril EP Impurity B; Anastrozole EP Impurity F | C₇H₈O₃S • x(H