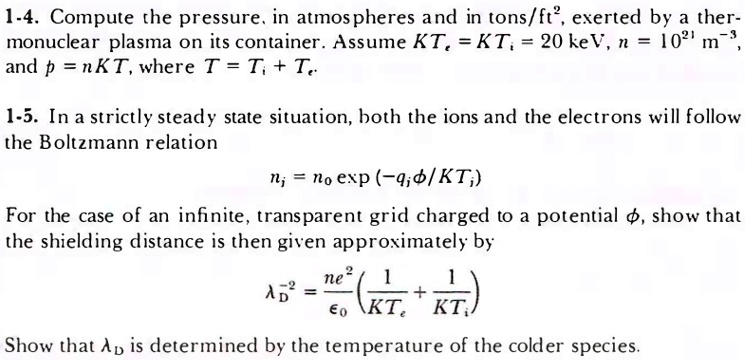
SOLVED: 1-4. Compute the pressure: in atmospheres and in tons/ft?, exerted by a ther monuclear plasma on its container Assume KT, KT; 20 keV, 102' and p nKT, where T = T; +

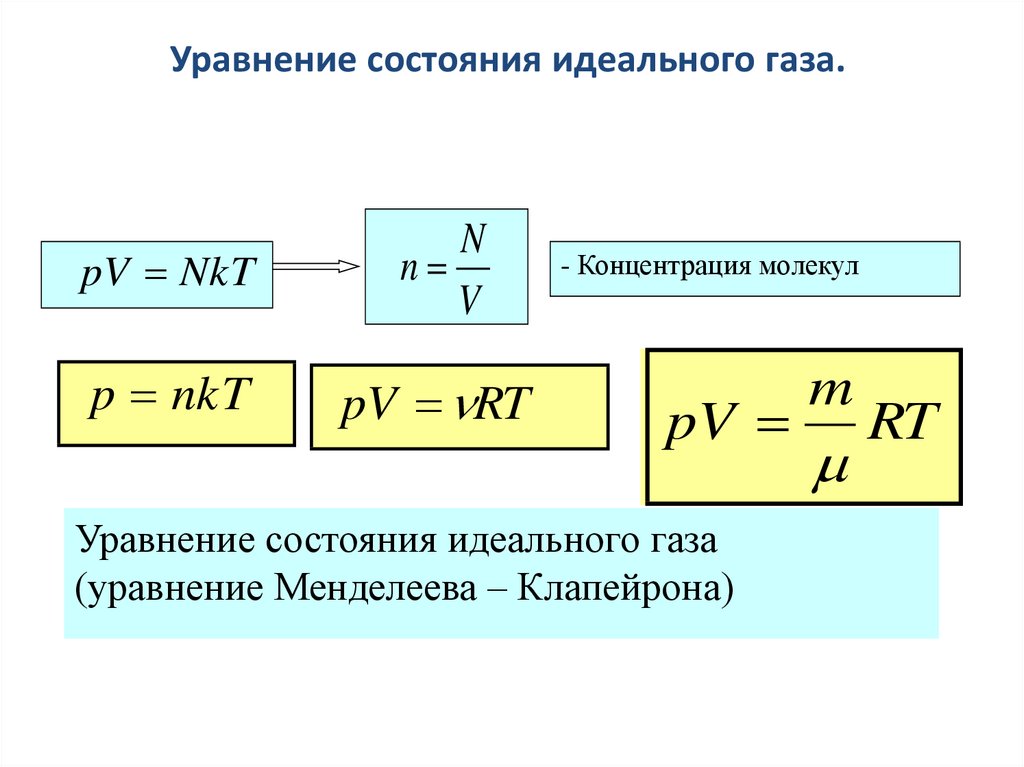
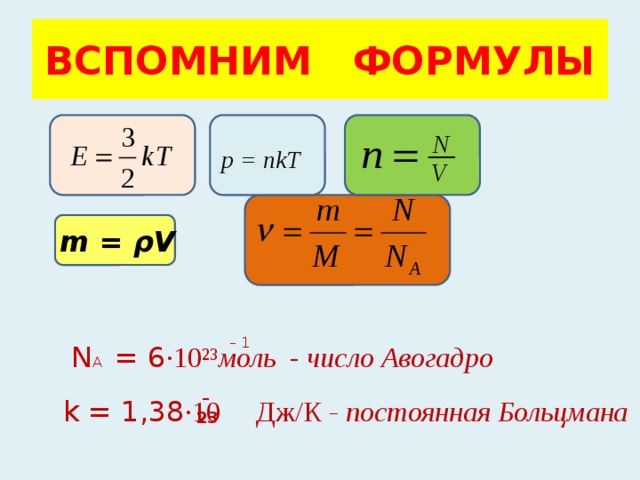
Презентация на тему: "Уравнение состояния R - универсальная газовая постоянная p=nkT.". Скачать бесплатно и без регистрации.

SOLVED: Recall the integrals: f FIn x + Constant OR dx =In X2 In X1 = In- Use this expression to calculate Wby dV when the ideal gas law pV = NkT

Ответы Mail.ru: Ребят, помогите решить задачу по физике, давление идеального газа. Задача-2 Вариант-1

SOLVED: Formula to predict the particles in the container is PV-NkT N= PVIkT Given P-47.3 atm V= 10.4 nm = 04x 10-26 T=300 K= 1.38x10-23 N= 2 (47.3)(1.04x 10-26 N = (1.38x10-23)(300) N= 532 mol