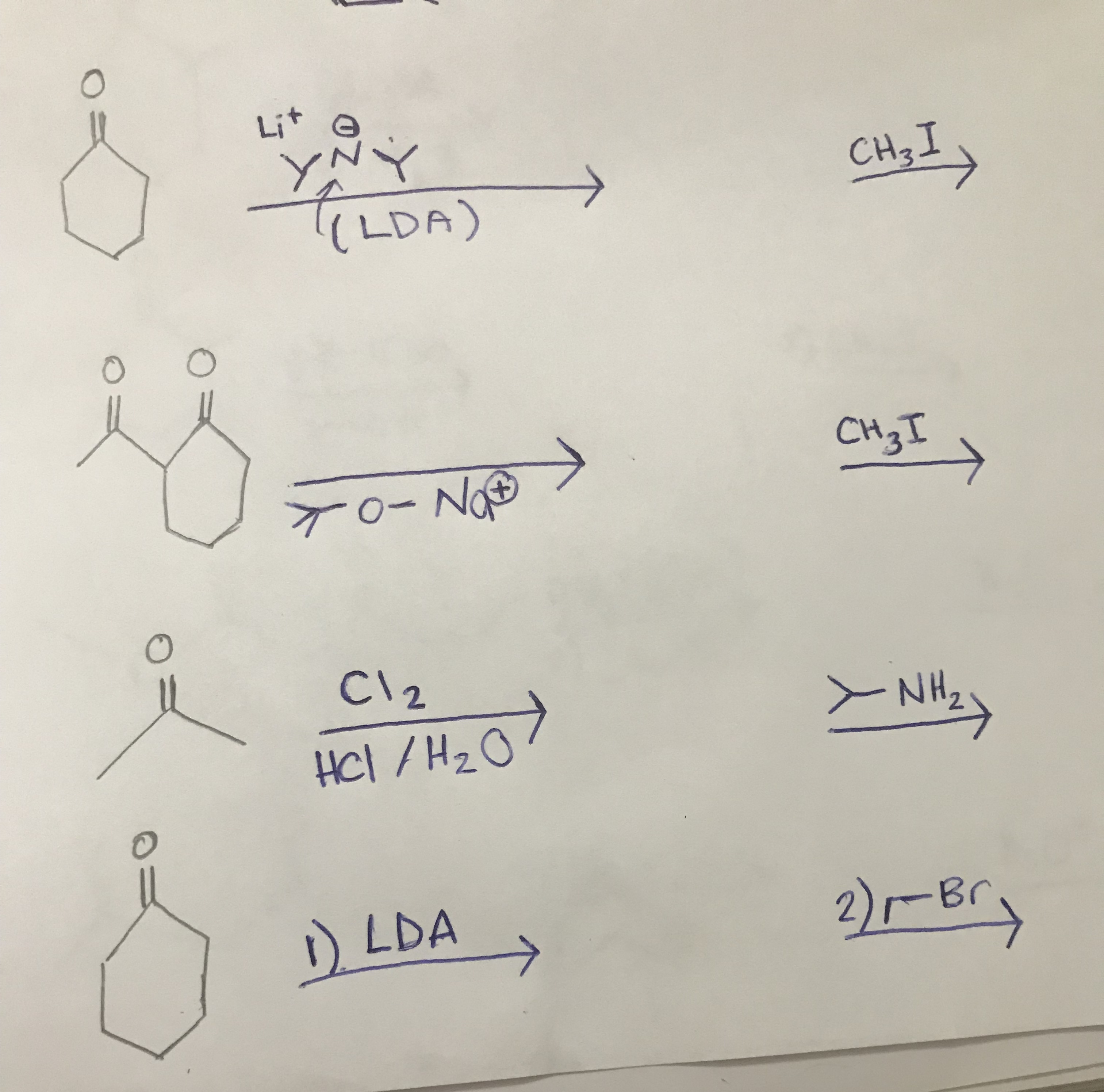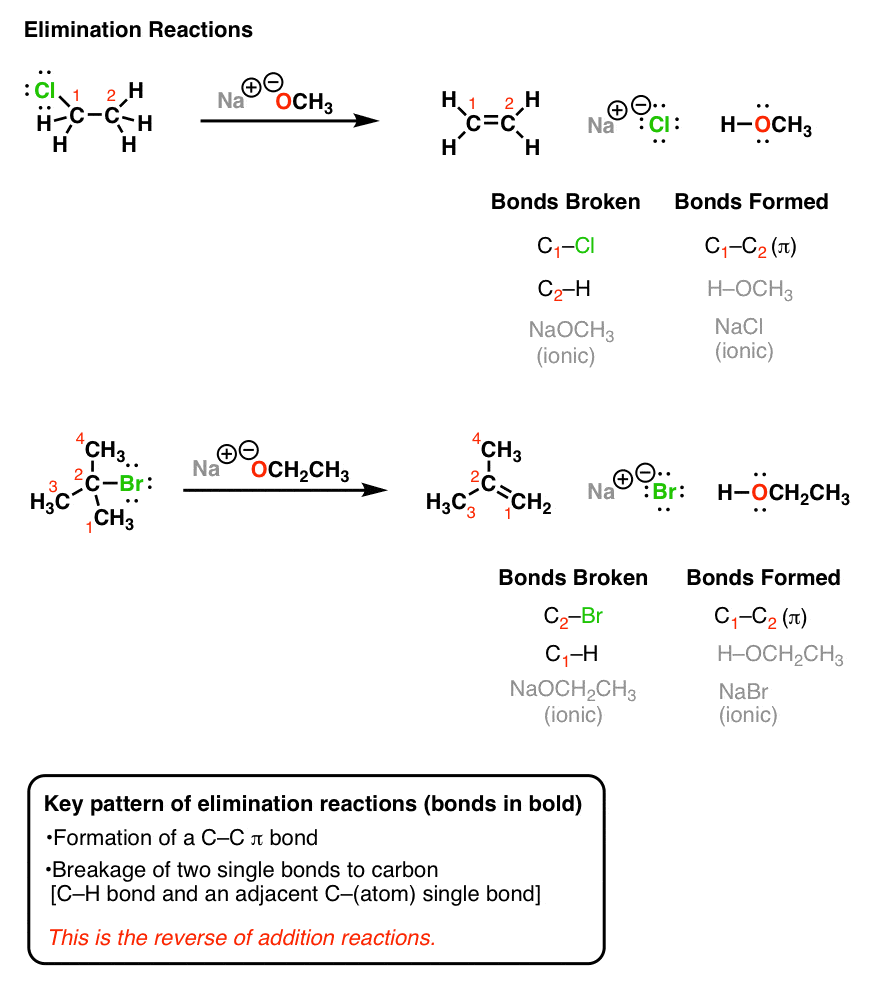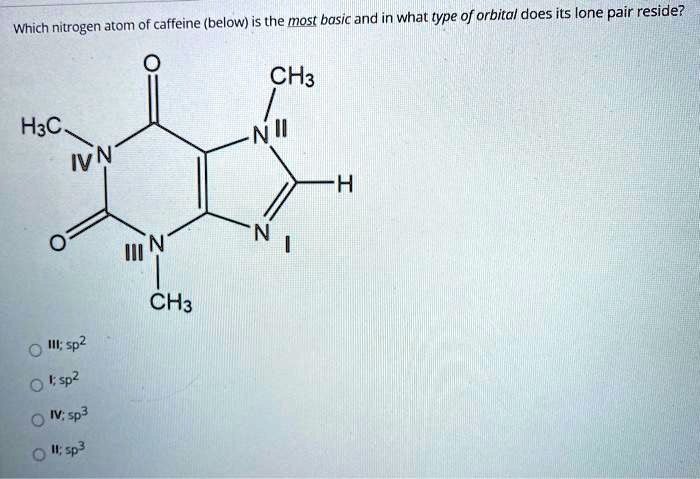
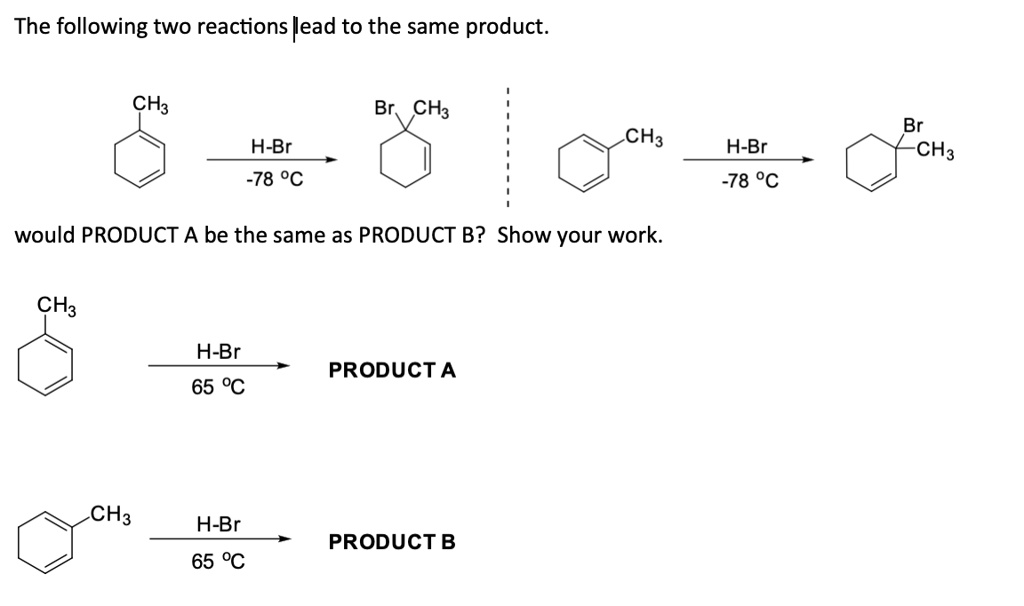
SOLVED: (below) is the most basic and in what type of orbital does its lone pair reside? Which nitrogen atom of caffeine CH3 H3C IVN CH3 I; 5p2 0 !sp2 0 I;
![Identify the product/s in the following reaction : 3CH3 CH = CH2 BH3 X H2 O2 / OH^ - [ Product/s + H3 BO3] Identify the product/s in the following reaction : 3CH3 CH = CH2 BH3 X H2 O2 / OH^ - [ Product/s + H3 BO3]](https://dwes9vv9u0550.cloudfront.net/images/4435926/c62057bd-b8cd-4e50-8a89-d07a45055ed1.jpg)
Identify the product/s in the following reaction : 3CH3 CH = CH2 BH3 X H2 O2 / OH^ - [ Product/s + H3 BO3]

Give reasons for the following: (CH3)3C–O–CH3 on reaction with HI gives (CH3 )3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I. - Chemistry | Shaalaa.com

Infrared Spectra of (Z)- and (E)-•C2H3C(CH3)I Radicals Produced upon Photodissociation of (Z)- and (E)-(CH2I)HC═C(CH3)I in Solid para-Hydrogen | The Journal of Physical Chemistry A
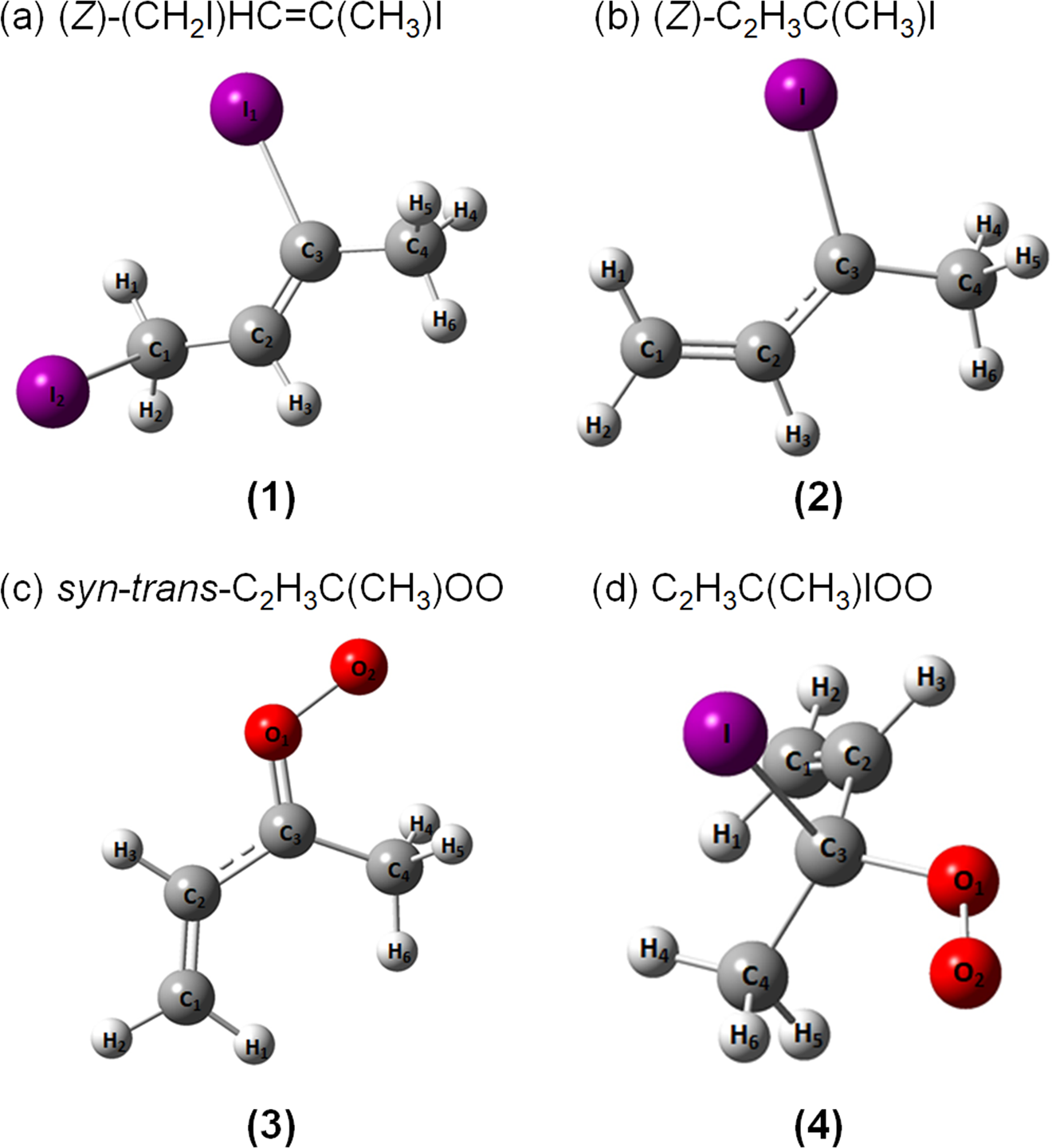
Infrared characterization of formation and resonance stabilization of the Criegee intermediate methyl vinyl ketone oxide | Communications Chemistry
CH3 C(CH3)2 CH2 OH + conc. H2SO4 > Alkene (major).Identify alkene Options: CH3 – CH2 – CH = CH2 CH3 – CH = C(CH3)2 (CH3)2C = C(CH3)2 CH3 CH=C CH3 I CH3

Write reactions of the final alkylation product of aniline with excess of methyl iodide in the p... - YouTube
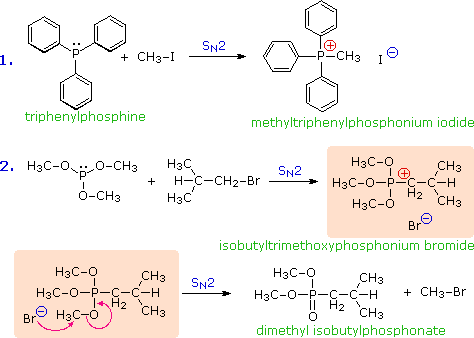
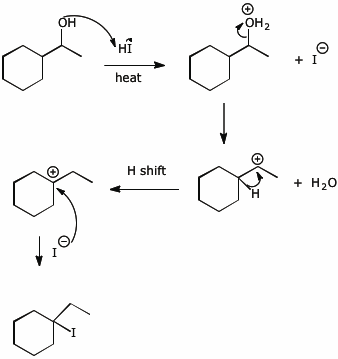

![Q115AE Iodomethane (CH3I) is a commonly... [FREE SOLUTION] | StudySmarter Q115AE Iodomethane (CH3I) is a commonly... [FREE SOLUTION] | StudySmarter](https://studysmarter-mediafiles.s3.amazonaws.com/media/textbook-exercise-images/image_XrFFo2b.png?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIA4OLDUDE42UZHAIET%2F20230531%2Feu-central-1%2Fs3%2Faws4_request&X-Amz-Date=20230531T004329Z&X-Amz-Expires=90000&X-Amz-SignedHeaders=host&X-Amz-Signature=197c9d23c566104573df19748cbfcaee641c488a94d12bcf148a6836dd8a02f7)