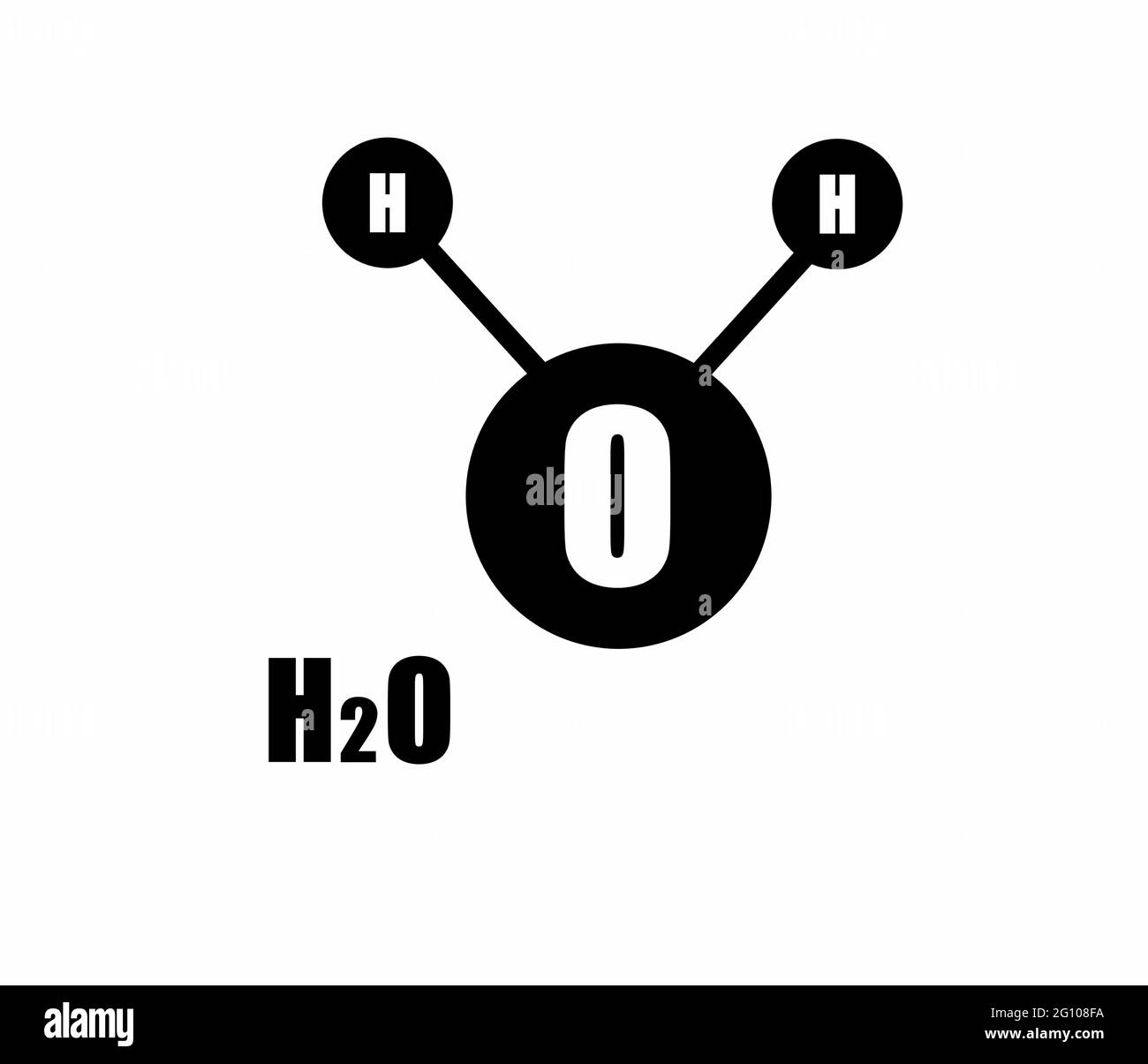
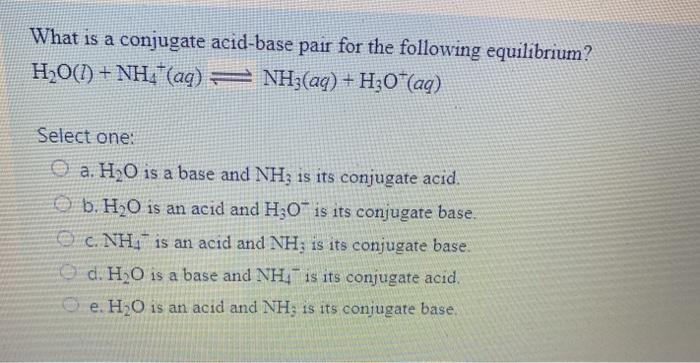![Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/jacs.8b11389/asset/images/medium/ja-2018-113892_0001.gif)
Cation Exchange Reactions in Layered Double Hydroxides Intercalated with Sulfate and Alkaline Cations (A(H2O)6)[M2+6Al3(OH)18(SO4)2]·6H2O (M2+ = Mn, Mg, Zn; A+ = Li, Na, K) | Journal of the American Chemical Society
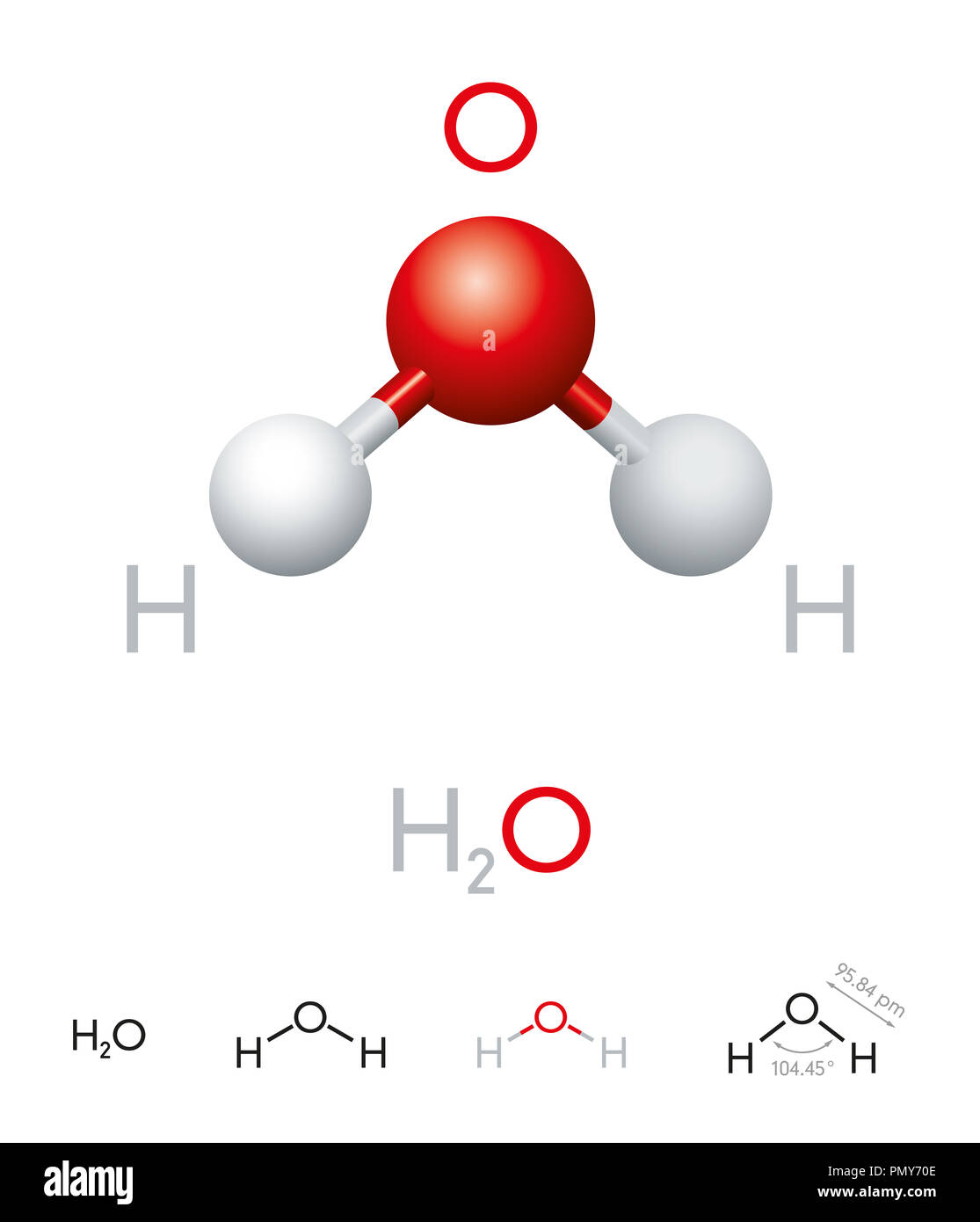
![Dissociation of H 2 O:H 2 O ↔ H + + OH - K w = a H+ a OH- a H2O Under dilute conditions: a i = [i] And a H2O = Dissociation of H 2 O:H 2 O ↔ H + + OH - K w = a H+ a OH- a H2O Under dilute conditions: a i = [i] And a H2O =](https://slideplayer.com/3401214/17/images/slide_1.jpg)
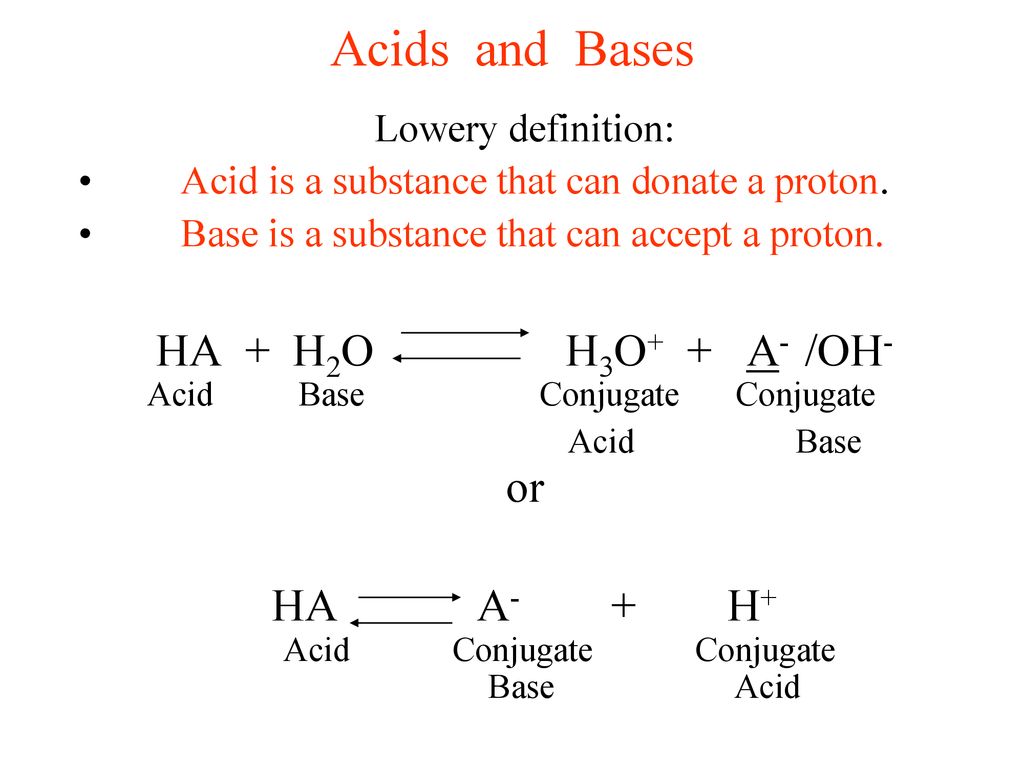
Dissociation of H 2 O:H 2 O ↔ H + + OH - K w = a H+ a OH- a H2O Under dilute conditions: a i = [i] And a H2O =
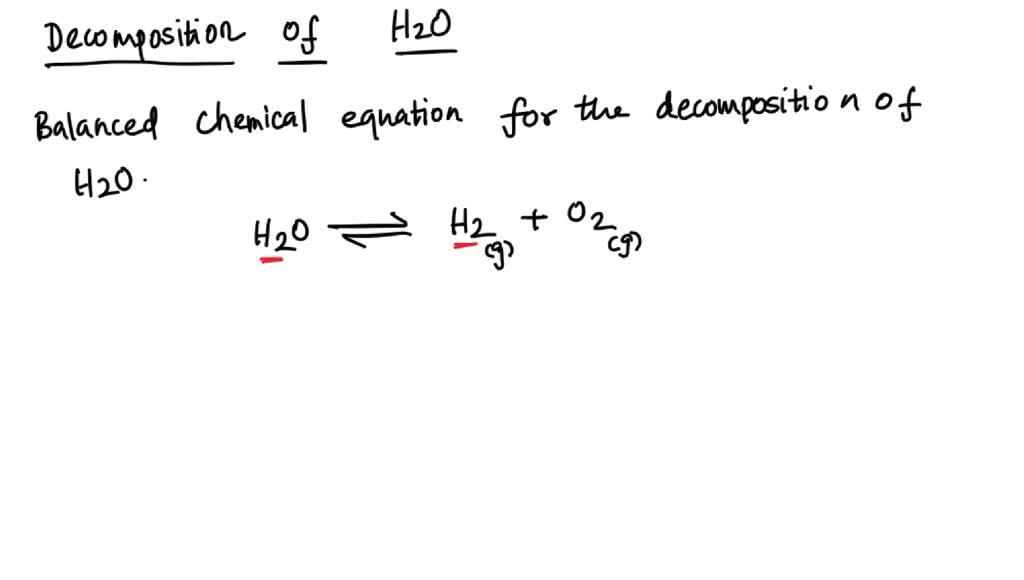
SOLVED: The decomposition of water into hydrogen gas H2 and oxygen gas O2 can be modeled by the balanced chemical equation A) H2 + O2 → H2O B) H2O → H2 +

SOLVED: In the following reaction: NH4+ + H2O = NH3 + H3O+ A) H2O is a base and NH3 is its conjugate acid B) NH4+ is an acid and H20 is its

SOLVED: Which of the following reactions is a hydrolysis reaction? 68) A) A B + C+ H2O B) A + B C + H2O C) A + B + H2O C D) A + H2O B + C
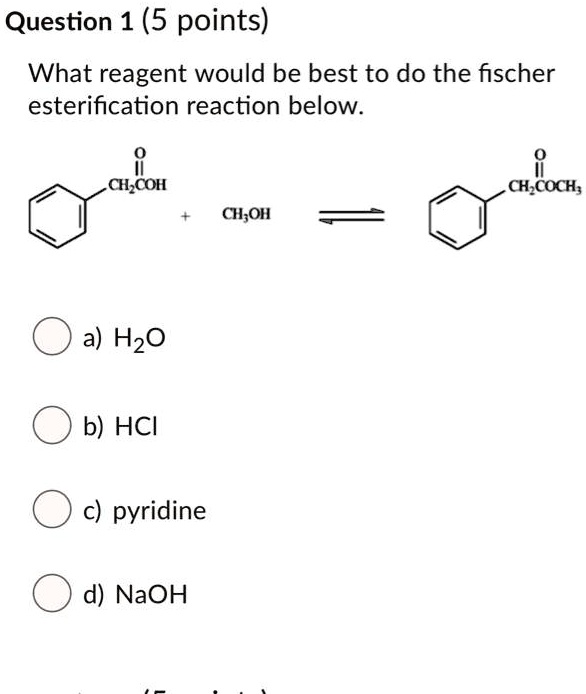
SOLVED: Question 1 (5 points) What reagent would be best to do the fischer esterification reaction below: CH,COH CH,COCH; CH;OH a) H2O b) HCI c) pyridine d) NaOH
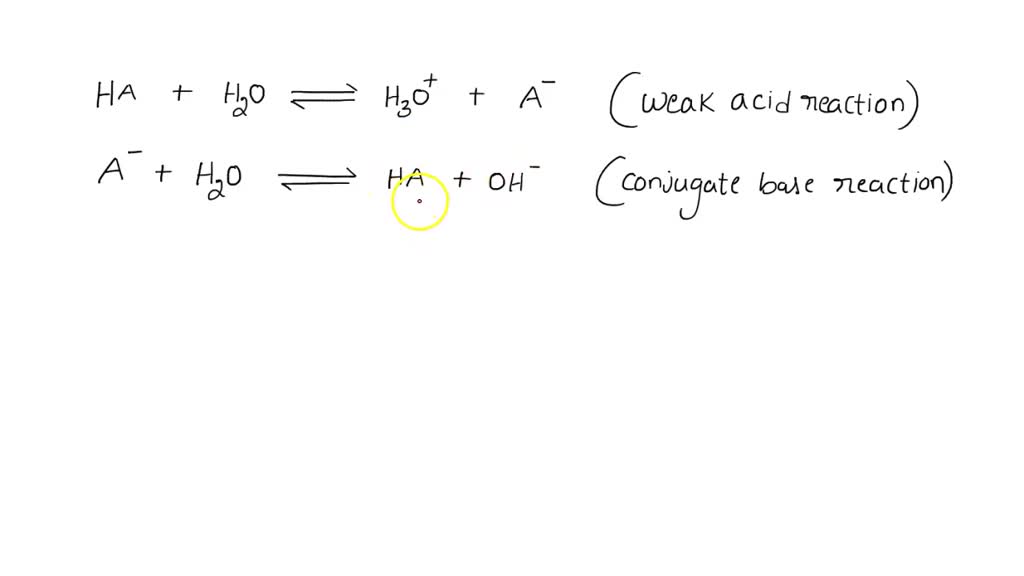
SOLVED: Consider the weak acid reaction: HA + H2O <-> H3O+ + A-1 and the reaction of its conjugate base: A-1 + H2O <-> HA + OH-1 Add the weak acid reaction